Publications
From animal and in vitro models to human cortical development & disease
Single-cell genotyping and transcriptomic profiling of mosaic focal cortical dysplasia (Baldassari, Klingler, et al., Nat Neurosci 2025)
Collaboration with Stéphanie Baulac and Sara Baldassari: Focal cortical dysplasia-linked epilepsy is more complex than expected – somatic mTOR-activating mutations affect multiple cell lineages, yet only a fraction become cytomegalic.
Transcriptional linkage analysis with in vivo AAV-Perturb-seq (Santinha, et al., Nature 2023)
In this collaboration with Randall Platt’s lab, we used AAV-Perturb-seq to dissect the transcriptional phenotypic landscape underlying 22q11.2 deletion syndrome in adult mouse prefrontal cortex.
Mapping the molecular and cellular complexity of cortical malformations (Klingler, Francis, Jabaudon & Cappello, Science 2021)
Cortical malformations cause various intellectual and/or motor disabilities often associated with seizures.
Here, we review how abnormal molecular and cellular developmental processes can lead to these malformations.
We further release an open access and user-friendly website, in which the expression of genes can be visualized across development along neuron differentiation, and compared between species (mouse vs. human) and conditions (in vivo vs. in vitro): http://www.humous.org/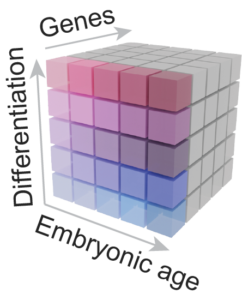
Emergence of cortical projection neuron diversity
Intrinsic and Input‐dependent Development of Cortical Neuron Types (Klingler, Roig Puiggros & Jabaudon, Wiley book chapter 2023)
In this chapter from the book Neocortical Neurogenesis in Development and Evolution, edited by Wieland Huttner, we summarize the intrinsic and extrinsic mechanisms allowing the emergence of cortical areas, and focus on how specific cell types (from progenitors to different types of excitatory and inhibitory neurons) have distinct susceptibilities to input during precise developmental windows.
Temporal controls over cortical projection neuron fate diversity (Klingler, Curr Opin in Neurobiol 2023)
During development, cortical projection neurons are sequentially produced and assemble into circuits. Cortical projection neurons are heterogeneous in terms of birthdate, layer position, molecular identity, connectivity and function. This diversity increases in evolutionarily most recent species, but when and how it emerges during corticogenesis is still debated. While time-locked expression of determinant genes and early stochasticity allow the production of different types of projection neurons, temporal differences in unfolding similar transcriptional programs, rather than fundamental differences in these programs, further account for anatomical variability between projection neuron subtypes and across species. Altogether, these mechanisms, discussed here, participate in increasing cortical projection neuron diversity.

Heterogeneous fates of simultaneously-born neurons in the cortical ventricular zone (Magrinelli et al., Sci Rep 2022)
Early-born deep layer (DL) cortical neurons comprise neurons projecting to subcortical structures (e.g. thalamus, spinal cord) and neurons projecting within the cortex (e.g. contralateral cortex). How does such diversity emerge during development?
Using both experimental and computational approaches, we show that immature and adult DL neurons have a high molecular heterogeneity. Interestingly, variable genes found in early-born immature neurons are genes which are normally expressed by late-born superficial layer neurons.
So, DL intracortically projecting neurons may have emerged from a variability in early transcriptional programs leading to the expression of genes normally expressed later during development, when superficial layer neurons are born.

Temporal controls over inter-areal cortical projection neuron fate diversity (Klingler et al., Nature 2021)
Interconnectivity between cortical areas is critical for sensorimotor processing. Although inter-areal cortical projection neurons are anatomically diverse, they are relatively homogeneous molecularly. How does this diversity of projections emerge during development?
Using ConnectID (i.e. MAPseq + scRNAseq) and in vivo functional approaches in the primary somatosensory cortex during postnatal development, we reveal a protracted unfolding of the molecular and functional differentiation of neurons projecting to the motor cortex (M), compared to those projecting to the secondary somatosensory cortex (S2).
We further identify SOX11, as a transcription factor whose dynamic expression is critical for the development of sensorimotor connectivity and subsequent exploratory behaviors.
Thus, dynamic differences in the expression of a largely generic set of genes, rather than fundamental differences in the identity of these genes, may account for the emergence of intra-type diversity of cortical neurons.
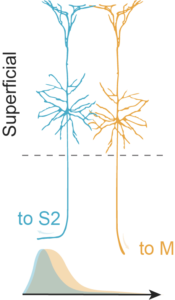
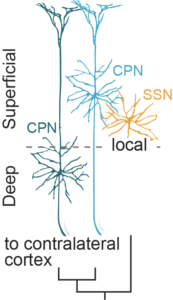
A translaminar genetic logic for the circuit identity of intracortically projecting neurons (Klingler et al., Curr Biol 2019)
In the primary somatosensory cortex, two main types of excitatory intracortically projecting neurons are found: layer (L)4 spiny stellate neurons (SSN) with short axon, locally projecting within the cortical column, and superficial and deep layer long-range projecting neurons, including callosally projecting neurons (CPN).
Using bulk RNAseq of SSN and retrogradely labeled CPN, we show that although far in layer position and date of birth, neurons projecting to similar places are molecularly closer than neurons with distinct projections.
This suggests that the molecular identities of cortical neurons more closely reflect their connectivity than their lineage relationship.
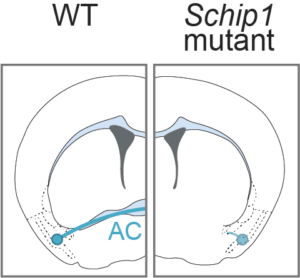
Development of inter-hemispheric fiber tracts
Inter-hemispheric connectivity is important to coordinate activity from each side of the brain. Among inter-hemispheric tracts, the anterior commissure (AC) connects the piriform cortices and amygdalae of both hemispheres.
Using transgenic mice, histology and in vitro assays, we show that SCHIP-1 is critical to guide AC axons to their target, notably through semaphorin-3F and EphB2 guidance cues.